Segmenting brain images with MAPER
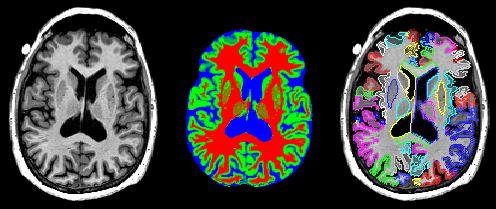
What it is
MAPER is a machine-learning method for anatomically segmenting magnetic resonance images of the human brain. The following paper describes the procedure:
Heckemann, R. A., Keihaninejad, S., Aljabar, P., Rueckert, D., Hajnal, J. V., Hammers, A., May 2010. Improving intersubject image registration using tissue-class information benefits robustness and accuracy of multi-atlas based anatomical segmentation. NeuroImage 51 (1), 221-227. http://dx.doi.org/10.1016/j.neuroimage.2010.01.072
MAPER is hosted at https://github.com/soundray/maper.
Table of Contents
- What it is
- Why segment brain images?
- Earlier work
- Atlases
- Work done with MAPER
- ADNI
- Connectivity-based subsegmentation of the thalamus
- Random-forest based classification
- Classification using PET and support vector machine
- Inflammation PET in traumatic brain injury
- Temporal lobe epilepsy
- EAST syndrome
- PET: increasing analytic accuracy
- PET: localizing focal uptake in tuberous sclerosis
- Alzheimer's disease: early amygdala involvement
- Alzheimer's disease: unexpected morphometric findings
- PET: region-based quantification
- Hippocampal volume in rheumatoid arthritis
- Parkinson's disease
- Graves’ disease
- Methods building on MAPER
- Future plans
Why segment brain images?
Anatomical brain image segmentation has a variety of uses:
- Quantitative anatomy: to describe anatomical phenotypes and individual variations in an intersubjective, reproducible manner
- Brain morphometry: to measure how diseases/conditions affect the shape and size of the structures that compose the brain
- Biomarker discovery and measurement: to obtain morphometric biomarkers for diagnosis, prognosis, and disease monitoring
- Intervention trials / drug testing: to monitor imaging biomarkers as surrogates for treatment response or disease progression
- Functional imaging support: to assign anatomical locations to functional activations in PET and fMRI.
MAPER processing requires preprocessing to eliminate non-brain tissue from the images (skull stripping or brain extraction). We developed Pincram, a highly accurate brain extraction method, by building on the same atlas-based principle as MAPER.
Earlier work
MAPER is based on earlier work on multi-atlas based segmentation:
Heckemann, R. A., Hajnal, J. V., Aljabar, P., Rueckert, D., Hammers, A., October 2006. Automatic anatomical brain MRI segmentation combining label propagation and decision fusion. NeuroImage 33 (1), 115-126. http://dx.doi.org/10.1016/j.neuroimage.2006.05.061
Atlases
Hammers_mith Adult Atlas Database
We primarily use a database of 30 MRI brain atlases of young healthy adults, partially available for download from www.brain-development.org (Resources - Atlases - Adult). The segmentation regions have been carefully prepared using detailed, validated protocols which are described in the following papers.
Baseline protocol (47 regions)
Hammers A, Allom R, Koepp MJ, Free S, Myers R, Lemieux L, Mitchell TN, Brooks DJ, Duncan JS, Aug. 2003. Three-dimensional maximum probability atlas of the human brain, with particular reference to the temporal lobe. Hum. Brain Mapp. 19 (4), 224-247. http://dx.doi.org/10.1002/hbm.10123
Extension to 83 regions
Gousias IS, Rueckert D, Heckemann RA, et al., 2008, Automatic segmentation of brain MRIs of 2-year-olds into 83 regions of interest, Neuroimage, Vol:40, ISSN:1053-8119, Pages:672-684. http://dx.doi.org/10.1016/j.neuroimage.2007.11.034
Extension to 95 regions
Insula subdivision
Faillenot I, Heckemann RA, Frot M, Hammers A. Macroanatomy and 3D probabilistic atlas of the human insula. Neuroimage. 2017 Apr 15;150:88-98. http://dx.doi.org/10.1016/j.neuroimage.2017.01.073
Parietal lobe subdivision
Wild HM, Heckemann RA, Studholme C, Hammers A. Gyri of the human parietal lobe: Volumes, spatial extents, automatic labelling, and probabilistic atlases. PLoS One. 2017 Aug 28;12(8):e0180866. http://dx.doi.org/10.1371/journal.pone.0180866
Extension to 120 regions
Work in progress. Among the new regions is the piriform cortex:
Steinbart D, Yaakub SN, Steinbrenner M, Guldin LS, Holtkamp M, Keller SS, Weber B, Rüber T, Heckemann RA, Ilyas-Feldmann M, Hammers A; Alzheimer’s Disease Neuroimaging Initiative. Automatic and manual segmentation of the piriform cortex: Method development and validation in patients with temporal lobe epilepsy and Alzheimer’s disease. Hum Brain Mapp. 2023 Jun 1;44(8):3196-3209. http://dx.doi.org/10.1002/hbm.26274
Other atlases
MAPER can be used with other atlases, for example the atlas database published in conjunction with the MICCAI 2012 Grand Challenge on Multi-Atlas Labeling (http://masiweb.vuse.vanderbilt.edu/workshop2012).
We compared MAPER and FreeSurfer using various atlas databases:
Yaakub SN, Heckemann RA, Keller SS, McGinnity CJ, Weber B, Hammers A. On brain atlas choice and automatic segmentation methods: a comparison of MAPER & FreeSurfer using three atlas databases. Sci Rep. 2020 Feb 18;10(1):2837. http://dx.doi.org/10.1038/s41598-020-57951-6
Work done with MAPER
ADNI
We processed the 996 baseline and screening images of ADNI (Alzheimer’s Disease Neuroimaging Initiative) with MAPER. A description of the data and volumetric analysis results appears in:
Heckemann RA, Keihaninejad S, Aljabar P, Gray KR, Nielsen C, Rueckert D, Hajnal JV, Hammers A, Alzheimer’s Disease Neuroimaging Initiative, 2011, Automatic morphometry in Alzheimer’s disease and mild cognitive impairment., Neuroimage, Vol:56, 1053-8119, Pages:2024-2037 http://dx.doi.org/10.1016/j.neuroimage.2011.03.014
The segmentations of the ADNI images are freely available for research purposes at the ADNI download site. After signing up as an ADNI participant, go through “Advanced Search”, select the “Post-Processed” radio button and enter “MAPER*” under “Series Description”.
More recently, we processed all available ADNI images (baseline, screening, and followup) with MALP-EM. These are also freely available – see section MALP-EM below.
Connectivity-based subsegmentation of the thalamus
For this work, we used diffusion tensor imaging to identify parts of the thalamus that connect to different cortical regions.
Traynor C, Heckemann RA, Hammers A, et al., 2010, Reproducibility of thalamic segmentation based on probabilistic tractography., Neuroimage, Vol:52, 1053-8119, Pages:69-85. http://dx.doi.org/10.1016/j.neuroimage.2010.04.024
Random-forest based classification
Gray KR, Aljabar P, Heckemann RA, Hammers A, Rueckert D; Alzheimer’s Disease Neuroimaging Initiative. Random forest-based similarity measures for multi-modal classification of Alzheimer’s disease. Neuroimage. 2013 Jan 15;65:167-75. http://dx.doi.org/10.1016/j.neuroimage.2012.09.065
Classification using PET and support vector machine
Gray KR, Wolz R, Heckemann RA, Aljabar P, Hammers A, Rueckert D; Alzheimer’s Disease Neuroimaging Initiative. Multi-region analysis of longitudinal FDG-PET for the classification of Alzheimer’s disease. Neuroimage. 2012 Mar;60(1):221-9. http://dx.doi.org/10.1016/j.neuroimage.2011.12.071
Inflammation PET in traumatic brain injury
This work is remarkable in that we found inflammatory activity long after the trauma event and remote from the injury site.
Ramlackhansingh AF, Brooks DJ, Greenwood RJ, Bose SK, Turkheimer FE, Kinnunen KM, Gentleman S, Heckemann RA, Gunanayagam K, Gelosa G, Sharp DJ, close, Inflammation after Trauma: Microglial Activation and Traumatic Brain Injury, Annals of Neurology, Vol:70, ISSN:0364-5134, Pages:374-383. http://dx.doi.org/10.1002/ana.22455
Temporal lobe epilepsy
This is a special one in that we showed involvement of the substantia nigra in temporal lobe epilepsy, which has not been documented before.
Keihaninejad S, Heckemann RA, Gousias IS, Hajnal JV, Duncan JS, Aljabar P, Rueckert D, Hammers A, 2012, Classification and lateralization of temporal lobe epilepsies with and without hippocampal atrophy based on whole-brain automatic MRI segmentation., PLoS One, Vol:7, Pages:1932-6203. http://dx.doi.org/10.1371/journal.pone.0033096
EAST syndrome
A study of a rare genetic syndrome. First introduction of MAPER-based volumetric fingerprinting.
Cross JH, Arora R, Heckemann RA, Gunny R, Chong K, Carr L, Baldeweg T, Differ AM, Lench N, Varadkar S, Sirimanna T, Wassmer E, Hulton SA, Ognjanovic M, Ramesh V, Feather S, Kleta R, Hammers A, Bockenhauer D, 2013, Neurological features of epilepsy, ataxia, sensorineural deafness, tubulopathy syndrome. Dev Med Child Neurol, Vol:55, 0012-1622, Pages:846-856. http://dx.doi.org/10.1111/dmcn.12171
PET: increasing analytic accuracy
Here, we used MAPER segmentations to correct partial volume effect in opioid receptor PET.
McGinnity CJ, Shidahara M, Feldmann M, Keihaninejad S, Barros DAR, Gousias IS, Duncan JS, Brooks DJ, Heckemann RA, Turkheimer FE, Hammers A, Koepp MJ, 2013, Quantification of opioid receptor availability following spontaneous epileptic seizures: Correction of [C-11]diprenorphine PET data for the partial-volume effect. Neuroimage 79:72-80. http://dx.doi.org/10.1016/j.neuroimage.2013.04.015
PET: localizing focal uptake in tuberous sclerosis
Regional analysis of PET enables anatomical location classification of tubers in the brain.
Rubi S, Costes N, Heckemann RA, Bouvard S, Hammers A, Marti Fuster B, Ostrowsky K, Montavont A, Jung J, Setoain X, Catenoix H, Hino K, Liger F, Le Bars D, Ryvlin P. Positron emission tomography with alpha-[11C]methyl-L-tryptophan in tuberous sclerosis complex-related epilepsy. Epilepsia. 2013 Dec;54(12):2143-50. http://dx.doi.org/10.1111/epi.12412
Alzheimer's disease: early amygdala involvement
In this work, we showed that amygdala atrophy characterizes early disease even better than hippocampal atrophy.
Klein-Koerkamp Y, Heckemann RA, Ramdeen KT, Moreaud O, Keignart S, Krainik A, Hammers A, Baciu M, Hot P; Alzheimer’s Disease Neuroimaging Initiative. Amygdalar atrophy in early Alzheimer's disease. Curr Alzheimer Res. 2014 Mar;11(3):239-52. http://dx.doi.org/10.2174/1567205011666140131123653
Alzheimer's disease: unexpected morphometric findings
For regions known to be involved in recognizing facial expressions of emotions, we made a surprise discovery: functional impairment is correlated with region size in early Alzheimer's. We had expected that recognition deficits would correlate with regional atrophy, but our results strongly indicate the opposite, at least in early disease.
Sapey-Triomphe LA, Heckemann RA, Boublay N, Dorey JM, Hénaff MA, Rouch I, Padovan C, Hammers A, Krolak-Salmon P; Alzheimer's Disease Neuroimaging Initiative. Neuroanatomical Correlates of Recognizing Face Expressions in Mild Stages of Alzheimer's Disease. PLoS One. 2015 Dec 16;10(12):e0143586. http://dx.doi.org/10.1371%2Fjournal.pone.0143586
PET: region-based quantification
Extensive evaluation of quantification strategies for a new cannabinoid receptor tracer, including region-based analysis.
Riano Barros DA, McGinnity CJ, Rosso L, Heckemann RA, Howes OD, Brooks DJ, Duncan JS, Turkheimer FE, Koepp MJ, Hammers A. Test-retest reproducibility of cannabinoid-receptor type 1 availability quantified with the PET ligand [11C]MePPEP. Neuroimage. 2014 Aug 15;97:151-62. http://dx.doi.org/10.1016/j.neuroimage.2014.04.020
Hippocampal volume in rheumatoid arthritis
In a mouse model of rheumatoid arthritis, we documented hippocampal changes correlating with disease markers. In pilot MR data on humans with rheumatoid arthritis, we found corresponding changes: hippocampal size was correlated with disease signs and symptoms.
Andersson KME, Wasén K, Juzokaite L, Leifsdottir L, Erlandsson MC, Silfverswärd ST, Stokowska A, Pekna M, Pekny M, Olmarker K, Heckemann RA, Kalm M, Bokarewa MI. Inflammation in the hippocampus affects IGF1 receptor signaling and contributes to neurological sequelae in rheumatoid arthritis. Proc Natl Acad Sci USA published ahead of print December 3, 2018. https://doi.org/10.1073/pnas.1810553115
Parkinson's disease
In early-stage Parkinson's disease, we showed correlations between limbic abnormalities and disease signs and symptoms.
Prange S, Metereau E, Maillet A, Lhommée E, Klinger H, Pelissier P, Ibarrola D, Heckemann RA, Castrioto A, Tremblay L, Sgambato V, Broussolle E, Krack P, Thobois S. Early limbic microstructural alterations in apathy and depression in de novo Parkinson's disease. Mov Disord. 2019 Jul 15. doi: http://dx.doi.org/10.1002/mds.27793
Graves’ disease
Holmberg M, Malmgren H, Heckemann RA, Johansson B, Klasson N, Olsson E, Skau S, Starck G, Filipsson Nyström H. A Longitudinal Study of Medial Temporal Lobe Volumes in Graves Disease. J Clin Endocrinol Metab. 2022 Mar 24;107(4):1040-1052. http://dx.doi.org/10.1210/clinem/dgab808
Methods building on MAPER
MALP-EM
Professor Christian Ledig developed MALP-EM. Thanks to an advanced label fusion process, MALP-EM achieves superior accuracy and robustness.
This paper demonstrates MALP-EM's strengths on images showing traumatic brain injury.
Ledig C, Heckemann RA, Hammers A, Lopez JC, Newcombe VF, Makropoulos A, Lötjönen J, Menon DK, Rueckert D. Robust whole-brain segmentation: application to traumatic brain injury. Med Image Anal. 2015 Apr;21(1):40-58. http://dx.doi.org/10.1016/j.media.2014.12.003
Here we discuss MALP-EM's application to all available ADNI images.
Ledig C, Schuh A, Guerrero R, Heckemann RA, Rueckert D. Structural brain imaging in Alzheimer's disease and mild cognitive impairment: biomarker analysis and shared morphometry database. Sci Rep. 2018 Jul 26;8(1):11258. http://dx.doi.org/10.1038/s41598-018-29295-9
Data are available at http://doi.org/10.12751/g-node.aa605a
DrawEM
DrawEM, a multi-atlas method for neonatal MR image segmentation has been developed by Dr. Antonios Makropoulos building on MAPER.
Makropoulos A, Gousias IS, Ledig C, Aljabar P, Serag A, Hajnal JV, Edwards AD, Counsell SJ, Rueckert D. Automatic whole brain MRI segmentation of the developing neonatal brain. IEEE Trans Med Imaging. 2014 Sep;33(9):1818-31. http://dx.doi.org/10.1109/TMI.2014.2322280
Selection of work using DrawEM:
Hellström W, Hortensius LM, Löfqvist C, Hellgren G, Tataranno ML, Ley D, Benders MJNL, Hellström A, Björkman-Burtscher IM, Heckemann RA, Sävman K. Postnatal serum IGF-1 levels associate with brain volumes at term in extremely preterm infants. Pediatr Res. 2023 Feb;93(3):666-674. http://dx.doi.org/10.1038/s41390-022-02134-4
Hortensius LM, Hellström W, Sävman K, Heckemann RA, Björkman-Burtscher IM, Groenendaal F, Andersson MX, Nilsson AK, Tataranno ML, van Elburg RM, Hellström A, Benders MJNL. Serum docosahexaenoic acid levels are associated with brain volumes in extremely preterm born infants. Pediatr Res. 2021 Dec;90(6):1177-1185. http://dx.doi.org/10.1038/s41390-021-01645-w
Future plans
Several further projects that use MAPER are underway.
Rolf Heckemann
Email: rolf dot heckemann at gu dot se